N-Ethyl-2-phenylpropan-1-amine
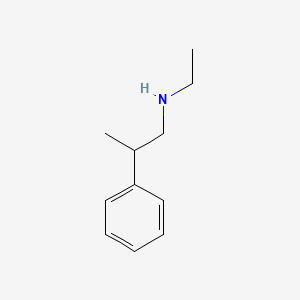
Synonyms: N-ethyl-Beta-Methylphenylethylamine
CID 15788198
Years ago the supplement industry released Beta-MethylPhenylethylamine (b-methyl-PEA) in an attempt to enhance the effects of phenylethylamine without impinging on the Federal Analog Act (FAA). These guidelines were developed to restrict the production of designer amphetamines through chemical manipulation. B-methyl-PEA was able to side step these guidelines, and since it was easily isolatable in various plants, its legality was rarely in question. Unfortunately, the same chemical nature which made it easily side step FAA also made it inert at even large doses. As Shulgin determined decades ago, a beta-methyl group is too far away from the nitrogen to sterically protect against MAO, and therefore the differences between regular PEA and b-Methyl-PEA are negligible.
N-ethyl-Beta-Methylphenylethylamine, on the other hand, possesses a fairly bulky ethyl substituent coming off the nitrogen. This substituent should protect the nitrogen from rapid MAO deamination, and therefore prolong the drugs half life significantly (See: Amphetamine vs. N-ethylamphetamine). Pharmacodynamically, this drug will act no different then regular PEA, although its longevity will enhance its catecholamine "releasing" properties, especially with acute supplementation. Similarly, the N-ethyl group will also increase the compounds amphipathism, which may expedite CNS penetration.
N-Benzyl-2-phenethylamine
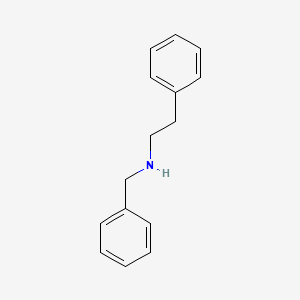
Synonyms: Benethamine, N-Benzyl-2-PEA
CID 65055
Extensive research with alpha-methylated derivatives of this compound (See: Benzphetamine, N-Benzylamphetamine) indicate that the main metabolite of N-Benzyl-2-PEA will be PEA. Similar to the compound previously discussed, the N-benzyl substituent will likely enjoy a longer half life than regular PEA. It should offer more CNS penetration due to the non-polarity of the benzyl moiety. The main difference between PEA and its N-benzyl derivative is that the latter may produce a local anesthetic effect relating to sodium channel inhibition. It may also possess weak sigma receptor proclivity in addition to theoretical 5-HT(2A) agonism.
In summary, these compounds may indeed be superior pharmacological derivatives of phenylethylamine, although vastly inferior to alpha-substituted analogues. N-benzyl-2-PEA is a constituent of the Erythropalum scandens species, and so is likely DSHEA compliant (1). Unfortuantely, no animal or human study exists, and the potential for negative health effects, although unlikely, are not excluded.
(1) Erythropalum scandens: http://japsonline.com/admin/php/uploads/283_pdf.pdf
Di-alkylated PEA derivatives have substantially less intrinsic activity vs. mono-alkylated PEA derivatives at catecholamine transporters (i.e. reuptake inhibition), although they are probably still able to stimulate norepinephrine release to some degree - probably the same as regular PEA or less. Alkylating the amine also increases the polarity between the amine and the benzene ring, which may increase CNS penetration. The half life of N,N-diethyl-PEA should also be longer then regular PEA (not hard to do since PEA is eliminated in 5-10 minutes without a MAOB inhibitor). And finally, PEA derivatives with bulky alkyl groups off the nitrogen sometimes possess an anesthetic effect.
ReplyDeleteYou seem very knowledgeable. Come join #reddit-nootropics :)
ReplyDeleteLooks like an interesting site.
ReplyDeleteWhat is known (or can be predicted) about the effects of phenethylamine derivatives on the TAAR? Or do we even know the SAR for TAAR agonists?
ReplyDeleteGreat blog! and very intersting article.
ReplyDeleteI have been researching into substituted PEA and was wondering if n, alpha diethyl PEA (allegedly found in DS Craze) could trigger a positive on a urine test?
What would the metabolic pathway be? 1-phenylbutan-2-amine --> norephedrine? But for that it would require a methyl group on the alpha instead of a ethyl... so I'm just a bit confused how the body would deal with this analog.
Cheers
ts a valuable content shared,would like to know more about it.
ReplyDeletePropiophenone
would you please tell me N-Benzyl-2-phenethylamine have what kind of therapeutic activity.???
ReplyDeletePhenethylamine belongs to a class of chemicals known as substituted Phenethylamines. The class consists of around 70+ compounds that cover a wide range of activities and applications and are known to be hallucinogens, appetite suppressants, central nervous system stimulants, antidepressants, entactogens, nasal decongestants and bronchodilators, vasopressors, and antiparkinsonian agents, among others.https://realchems.com/phenylethamines
ReplyDelete